
Validation & Compliance
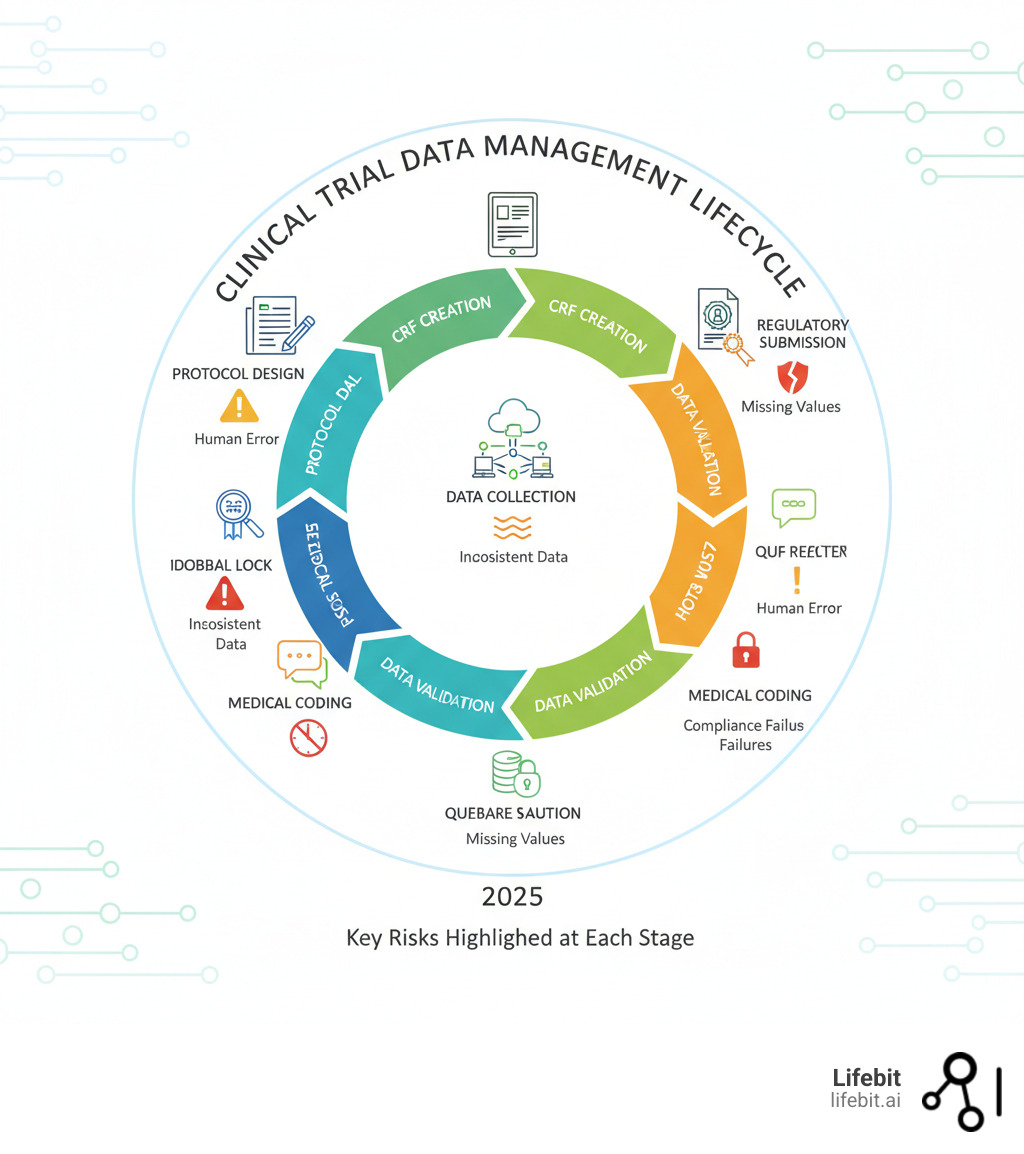
CDISC validation ensures datasets meet regulatory standards like SDTM and ADaM for submissions to FDA and PMDA. Pinnacle 21 software automates checks against business rules, identifying errors early to streamline compliance.
Validation Process
Run checks iteratively: start during data collection with CDASH-compliant CRFs, validate SDTM domains for structure and consistency, then ADaM for analysis readiness. Address errors by severity—critical issues block submissions—document resolutions in the reviewer's guide. FDA requires SDTM format; non-compliance delays reviews, while validated packages reduce costs and enable rapid assessments.

